أهمية الامتثال
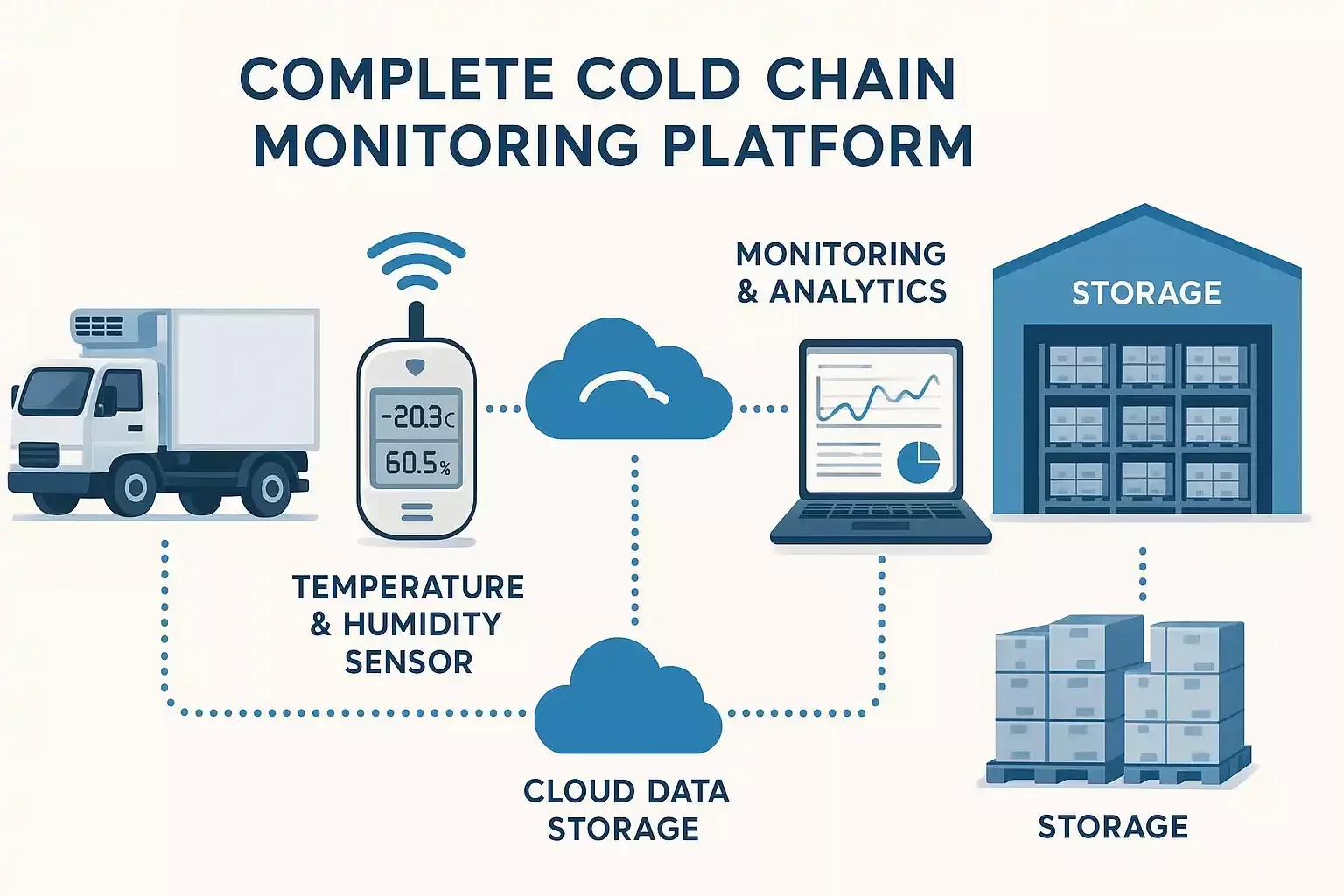
In regulated industries, data accuracy and traceability are critical. We deliver secure, auditable, and transparent data every step of the way.
5-Year Data Retention
Secure cloud archiving meets long-term pharma and food safety retention clauses.
Audit Trail
Fully traceable logs of every user action, alarm, and corrective measure.
Calibration
Factory-calibrated sensors with EN 13485 accuracy.
Zero Data Loss
Gateways buffer data during outages to ensure continuous records.
لمحة سريعة عن المعايير
Seemoto supports the most demanding regulatory frameworks across pharma, food, and logistics.
Compliance Standards in Detail
Each standard addressed with purpose-built monitoring, reporting, and validation capabilities.
Security
سلامة وأمن البيانات دون أي تنازلات
End-to-End Encryption
يتم تشفير البيانات من المستشعر إلى البوابة وطوال عملية النقل إلى السحابة، مما يمنع اعتراضها أو التلاعب بها.
Role-Based Access Control
تضمن أذونات المستخدم التفصيلية أن الموظفين المصرح لهم فقط هم من يمكنهم عرض البيانات أو تأكيد الإنذارات أو تعديل الإعدادات.
Immutable Audit Trails
Every system event including logins, alarm handling, and configuration changes is permanently logged and time-stamped.
Secure Cloud Retention
تلبية متطلبات الاحتفاظ طويل الأجل للبيانات وفقًا للوائح إدارة الغذاء والدواء الأمريكية (FDA) والاتحاد الأوروبي من خلال أرشفة البيانات تلقائيًا لمدة 5 سنوات.
موارد
موارد دعم الشركاء والتحقق من صحتهم
Compliance is a team effort. Seemoto empowers our partners and customers with ready-made validation toolkits and comprehensive training.
- Access our Partner Portal for IQ/OQ templates, user manuals, and calibration certificates
- Rely on our dedicated support team for technical and regulatory guidance during audits

الأسئلة الشائعة
Ready to Simplify Compliance?
Contact our team to learn how Seemoto can help you meet regulatory requirements with confidence.
